Chenchen Qu, Jeremy B Fein, Wenli Chen, Mingkai Ma, Peng Cai, Qiaoyun Huang. Mechanistic investigation and modeling of Cd immobilization by iron (hydr)oxide-humic acid coprecipitates, Journal of Hazardous Materials, 2021, 126603, https://doi.org/10.1016/j.jhazmat.2021.126603
Highlights
- •Cd stabilized by the freshly precipitated iron (hydr)oxide through adsorption.
- •Fresh iron (hydr)oxide has ~four times more sites for Cd than the pre-formed one.
- •HA promotes the precipitation of Fe clusters through ligand exchange reactions.
- •The majority of Cd was associated with iron (hydr)oxide in the composites.
- •Distribution of Cd can be predicted by site masking and Fe hydroxyl increment.
Abstract
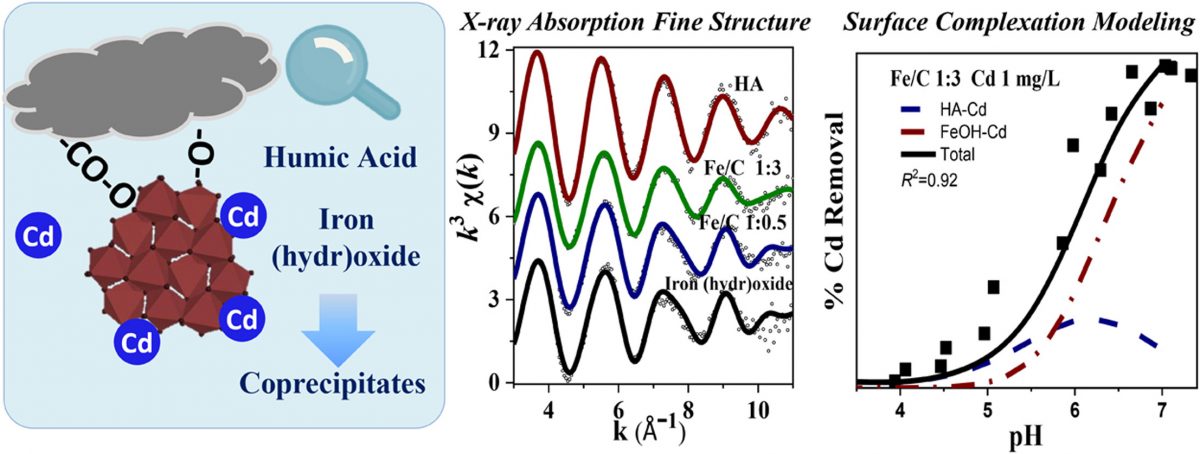
A molecular-scale understanding of aqueous metal adsorption onto humic acid-iron (hydr)oxide coprecipitates, and our ability to model these interactions, are lacking. Here, the molecular-scale mechanisms for Cd binding onto iron (hydr)oxide-humic acid (HA) composites were probed using X-ray absorption fine structure (XAFS) spectroscopy and surface complexation modeling (SCM). The immobilization of Cd in (hydr)oxide precipitation systems occurs predominantly through adsorption onto the freshly-formed (hydr)oxide nanoparticles, and SCM calculations suggest a specific surface area of 2,400 m2/g available for Cd. The solution and XAFS measurements indicate that HA promotes the precipitation of both Fe clusters and Fe-Cd associations mainly through ligand exchange reactions. Site masking reactions result in a dramatic blockage of functional sites on HA and ~45% migration of the adsorbed Cd to iron (hydr)oxide binding sites at high HA:Fe mass ratios. A composite model that accounts for both site masking between Fe ions and HA and the increase of Fe hydroxyl sites simulate the distribution of Cd in the composites reasonably well. Overall, this study demonstrates that the Fe clusters play an overriding role for heavy metal stabilization in coprecipitation systems, while HA promotes the immobilization of Cd by facilitating the flocculation and dispersion of Fe clusters.
全文链接:https://doi.org/10.1016/j.jhazmat.2021.126603